Haemodialysis Powder
Product Overview
Haemodialysis Powder is a dialysis concentrate powder used to prepare dialysate solution for haemodialysis treatment. It contains essential electrolytes such as sodium, potassium, calcium, magnesium, chloride, acetate, and bicarbonate to help maintain electrolyte balance in patients during dialysis.
The powder is dissolved in purified water using a dialysis powder mixing machine to produce concentrate solution for dialysis systems.
This product is economical, easy to transport, and allows flexible adjustment of electrolyte composition based on patient clinical requirements.
Feature:
- Made from high-purity, medical-grade chemicals
- Easy to prepare by mixing with water for dialysis solution
- Provides accurate and stable electrolyte composition
- Effectively removes metabolic waste from the body
- Helps maintain fluid, electrolyte, and acid-base balance
- Highly soluble, preventing blockage in the system
- Secure packaging to protect from moisture and contamination
- Compatible with most standard dialysis machines
Package Method:
- Haemodialysis Powder A: 10 bags / carton
- Haemodialysis Powder B: 20 bags / carton
![]()
![]()

To order, call: (+855) 093 265 939 (24/7 support)
Product Specifications
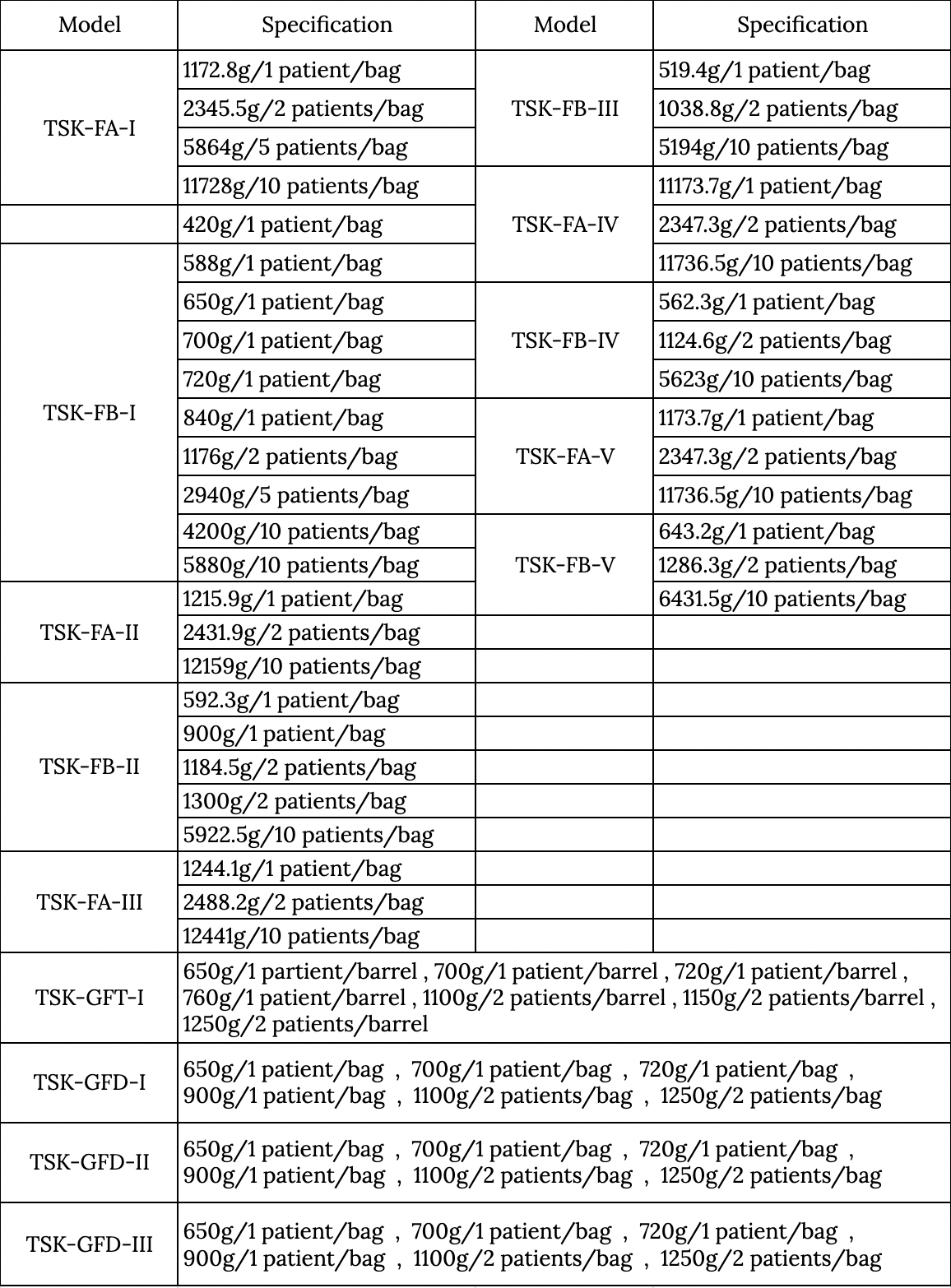
Applicable machine types: To be used with the appropriate three stream haemodialysis machine and bicarbonate concentrate. Applicable to the model which ion concentration is matches after dialysis be prepared (used so far).
Indications: To be use in a dialysis treatment for patients suffering from acute renal failure, chronic renal failure or acute intoxication with dialysable substances.
Scope of application: It is applicable to haemodialysis, Use with dialysis machine. (applicable to haemodialysis after the haemodialysis powder is formulated into solution, it is used in haemodialysis in comjuncation with comjuncation with haemodialysis machine.
Clinical benefit: The device is Used for treating kidney failure, uremia, anuresis and electrolyte disturbance caused by drug poisoning, the haemodialysis treatment can obviously improve the patient life quality.
Contraindications: Haemodialysis has no absolute contraindications. But to prevent accidents in dialysis, following should be listed as relative contraindications:
- Severe hemorrhage or anemia.
- Severe hypotension or shock
- Severe heart brain complications, such as distinct cardiomegalia accompanied with cardiac insufficiency, severe arrhythmia, severe hypertension or cerebrovascular disease
- Terminal uremia with inconvertible complications
- Uncontrolled diabetes.
- Severe infection
- Already suffering malignant diseases such as carcinoma, etc.
- within three days after major operation
- Old high risk patients, psychosis sufferers, uncooperative babies and infants
Notice:
- The product is a non-injection product, Please follow the doctor’s advice.
- Don’t use singly.
- Powder A and powder B should be used up once opened. Do not use if seals or container are damaged.
- Powder A prohibits the use of metal containers, weighing, mixing, storage allocation, powder A and powder B mix thoroughly before use. The operation of the specially adapted dialysis machine is restricted to persons who are trained in dialysis and who are completely familiar with the operating instrution stated in the operator manual of the corresponding dialysis machine.
- Please refer to the operating manual of dialysis machine to confirm the concentrated dialysis model, pH value and the mixture ratio.
- The formulation water of dialysis must conform to ISO 23500-3:2019 Water for haemodialysis and related therapies standard.
- Suggestion: Before use, please test the final dialysis fluid for one of the following parameters: conductivity, pH, osmotic pressure, sodium concentration, or chloride concentration.
- If additional electrolyte and buffer content is added to the dialysis fluid, in selecting the bicarbonate prescription, the physician should consider all sources of buffer delivered to the patient during the dialysis treatment, including the bicarbonate in the bicarbonate concentrate, the acetate, citrate or lactate in the acid concentrate which, when metabolized from bicarbonate. Also, the physician should consider the patient’s nutritional status, assessed by history, physical examination, anthropometrics, serum albumin and protein nitrogen appearance, since individuals whose metabolism results in a small acid load are at higher risk of developing metabolic alkalosis following treatment. To achieve the clinically required dialysis fluid composition in respect of electrolyte and buffer content, users should pay attention to ensure that the mix ratios used are appropriate and intended for the proportioning system or dialysis machine in use.
- The device is for single use, reuse of packaging materials will create cross infection, contamination etc.;
- After used, the device package shall be disposed according to local regulation as medical waste.
- The device shall be used strictly following the defined Formulation variables, unmatched use will create risks to patients.
- When being aware of any serious incident that has occurred in relation to the device, The user and/or patient should report it to the manufacturer and the competent authority of local countries.
- The disposal of products and package in accordance with hospital, administrative and/or local government policy.
Adverse reactions: Some undesirable effects related to dialysis can occur, for instance hypotension, nausea, vomiting and cramps.
Storage: well-sealed and put in drafty places, protected from direct sunshine and freezing, no mixed storage with objects which are poisonous, contaminative and have unpleasant odor. Save temperature +5°C~+30℃℃
Warnings:
Powder A: This acid concentrate product is for use as one component in mixing dialysis bath. This product contains citric acid. During dialysis, the dynamic of diffusion and concentration gradients prevent serum bicarbonate concentration from exceeding the dialysis bicarbonate concentration. The bicarbonate concentration of the dialysis is the bicarbonate setting on the dialysis machine, and is the bicarbonate dose prescribed by the physician. Prescription of insufficient bicarbonate may contribute to metabolic acidosis; excessive bicarbonate may contribute to metabolic alkalosis. Both conditions are associated with poor patient outcomes, including increased mortality risk.
Powder B: Please examine the shell and content before use. Never use it if breakage or pollution is found or suspected. Please refer to the IFU of dialysis machine. Do not overmix bicarbonate concentrate. Prescription of insufficient bicarbonate may contribute to metabolic acidosis; excessive bicarbonate may contribute to metabolic alkalosis.
Endotoxin: after the product is diluted with water used for endotoxin examination into dialysate, the endotoxin is less than 0.5 EU/ ml.
Microbe limit: Standard dialysis fluid shall contain a total viable microbial count of less than 100 CFU/ml
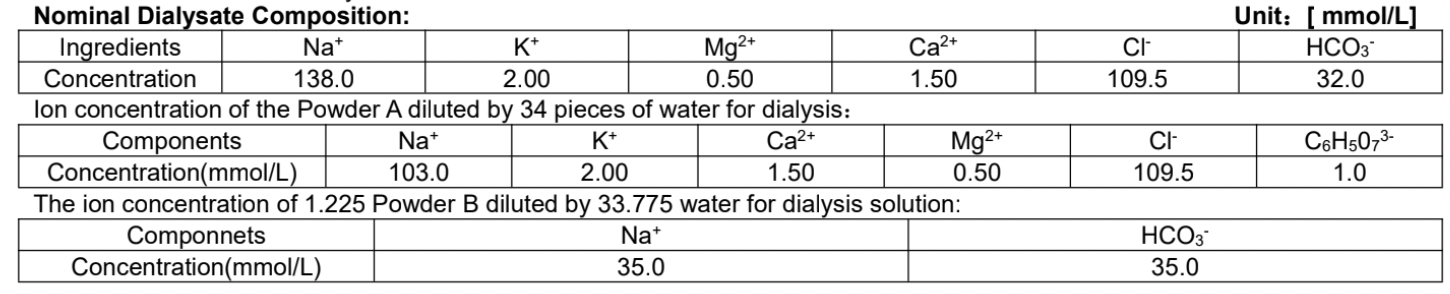
pH range: 7.0 – 7.6 (The pH range in this instruction is the result of test in laboratory. It is recommended to re-measure the value as per the requirement before clinical use.
Validity: Dialysis Powder A (TSK-FA-I) for 24 months; Dialysis Powder B (TSK-FB-I) for 24 months; Disposition method of medical waste: dispose as medical waste according to local laws.