A.V Fistula Needle
Product Description
Disposable A.V. Fistula Needle Sets (AVF Needle) are designed for vascular access during hemodialysis treatment. The needles are manufactured from high-quality stainless steel and medical-grade materials to ensure safe, stable, and efficient blood flow during dialysis procedures.
The needle features an ultra-thin wall and a triple-bevel design to minimize puncture resistance and improve patient comfort. Each needle set is sterilized with Ethylene Oxide (EO) and individually packaged for single use.
The product is suitable for use in dialysis centers, hospitals, and clinical dialysis treatments.
Features
- DEHP / DEHP-FREE
- BACK-EYE: Optimizing blood flow and minimizing the risk of aspiration into the vessel wall
- BLACK/ RED POINT: Mark the needle bevel
- WINGS: rotary or fixed
- STERILE: Ethylene Oxide (E.O.)
- MATERIAL: stainless steel SUS 304, medical grade plastic, latex free, non pyrogenic
- NEEDLE WALL: Ultra-thin, silicone-coated to prevent coagulation.
- 3 – BEVEL DESIGN: Less resistance during puncture, decrease patient pain.
Package Method: 50pcs/box, 10 boxes/Carton
![]()

![]()

To order, call: (+855) 093 265 939 (24/7 support)
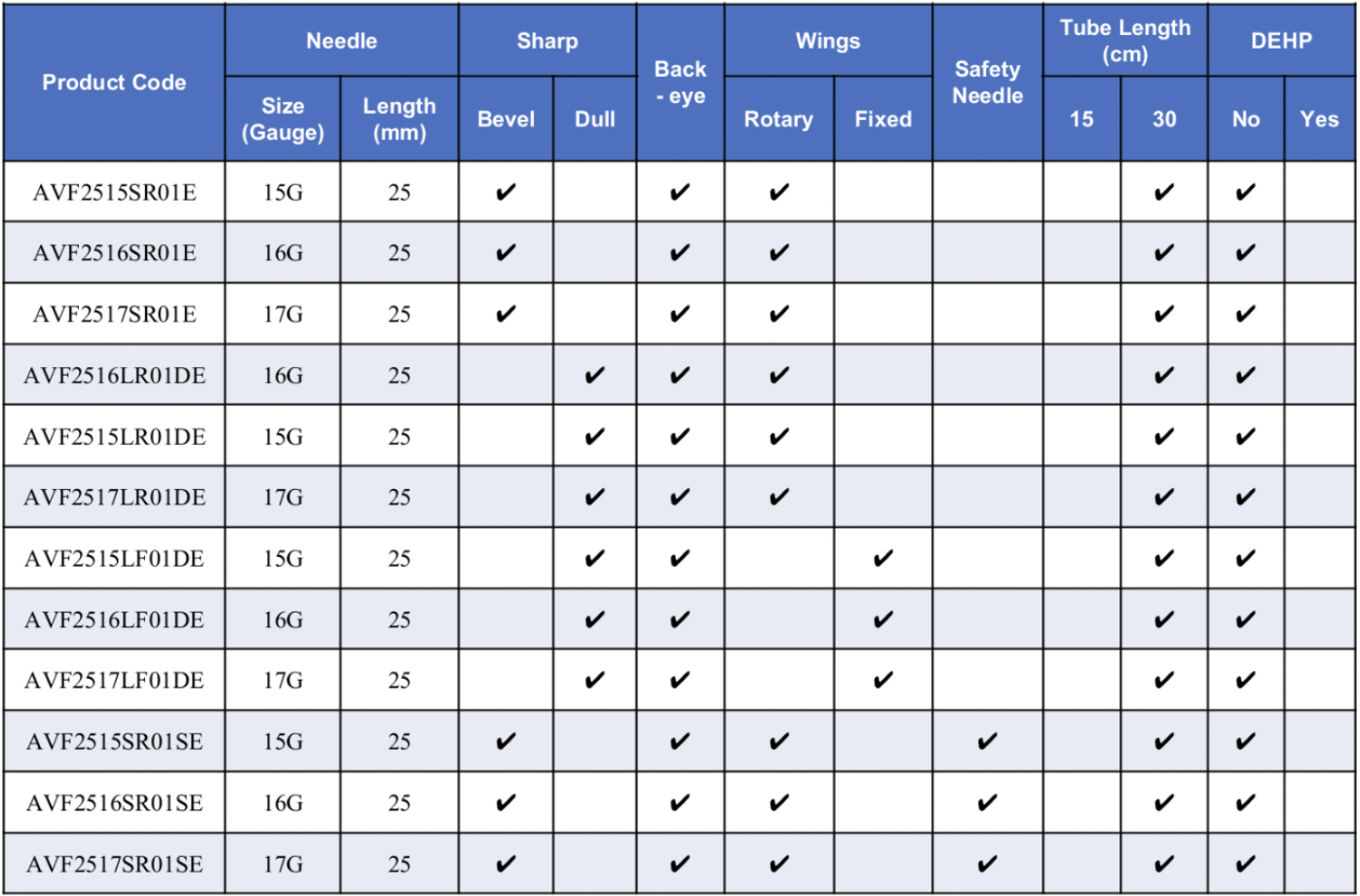
Needle Gauge & Blood Flow
- Gauge: 15G, Blood Flow Rate: 350-450ml/min, Color: Blue
- Gauge: 16G, Blood Flow Rate: 300-350ml/min, Color: Green
- Gauge: 17G, Blood Flow Rate: <300ml/min, Color: Red
Specifications
1. Material
The major components of this product are made from medical-grade PVC, PP, PE and other medical-grade macromolecule materials and stainless steel. And this product is free of latex.
2. Product configuration and Feature
AV Fistula needle set can be inserted to patient’s vein, and then connected to Hemodialysis bloodline tubing set. The tubing is soft, transparent, smooth and non-kink. The wing of the needle set is proper rigidity suitable for use. The on-off clamp is simple and convenient for use. The wing for 15G is Blue, 16G is Green, 17G is Red. The tip of the cannula is sharp to reduce the pain for the patient. The backeye is slender close to the cannula tip. It is designed with slippery prevention for smooth puncture.
3. Technical Performance
The products is fully sterilized by Ethylene Oxide gas, sterility and non-pyrogenicity.
4. Indication for use
This products is intended to be used as vein puncture for the hemodialysis treatment.
5.Usage method
- Take out the needle set from the pouch.
- Disinfect the skin where is intented to puncture.
- Screw out the cap for Female Luer Lock, pick out the cap for cannula, exclude all the air in tube with normal saline.
- Hold the wing, then puncture the vein. Clamp the tube once the blood goes into the tube.
- Fix the cannula on the puncture site by the medical adhesive tape.
- Connect with the bloodline tubing set.
- According to the above mentioned guidance, the same operation to the other needle set.
6. Transportation and storage
Please avoid crash or exposure to rain, snow or direct sunlight during transportation. Store it in 0°C~40°C, well-ventilated indoor place with relative humidity no more than 80%, without corrosive gases. Do not store it in warehouse with chemicals and moist articles.
7. Precautions in use
- Using this device should be supervised by a physician. Use aseptic technique throughout priming and treatment. The validity period of sterilization is three years. Please check the expiration date prior to use, to prevent contamination or infection, do not use any expired product.
- Do not use the product if the pouch is damaged.
- Open the pouch and pick out the AV Fistula needle set carefully.
- If the AV Fistula set can not be properly connected, or there is any fluid leakage or presence of air bubbles, treatment or readjustment should be performed by physician. In case no improvement is made, replace with another new AV Fistula needle set. Any abnormal condition should be properly treated under the direction of physician.
- This product is for single use only and reuse is strictly prohibited. Reprocessing of this product may lead to adverse patient reactions and/or device failure. It should be discarded according to laws and regulations relevant to disposal of infectious medical waste so as to prevent infection.
- All of the disinfectant used for this product have no special contraindications.
8. After sales service
Please keep the original packing for any investigation on product quality.
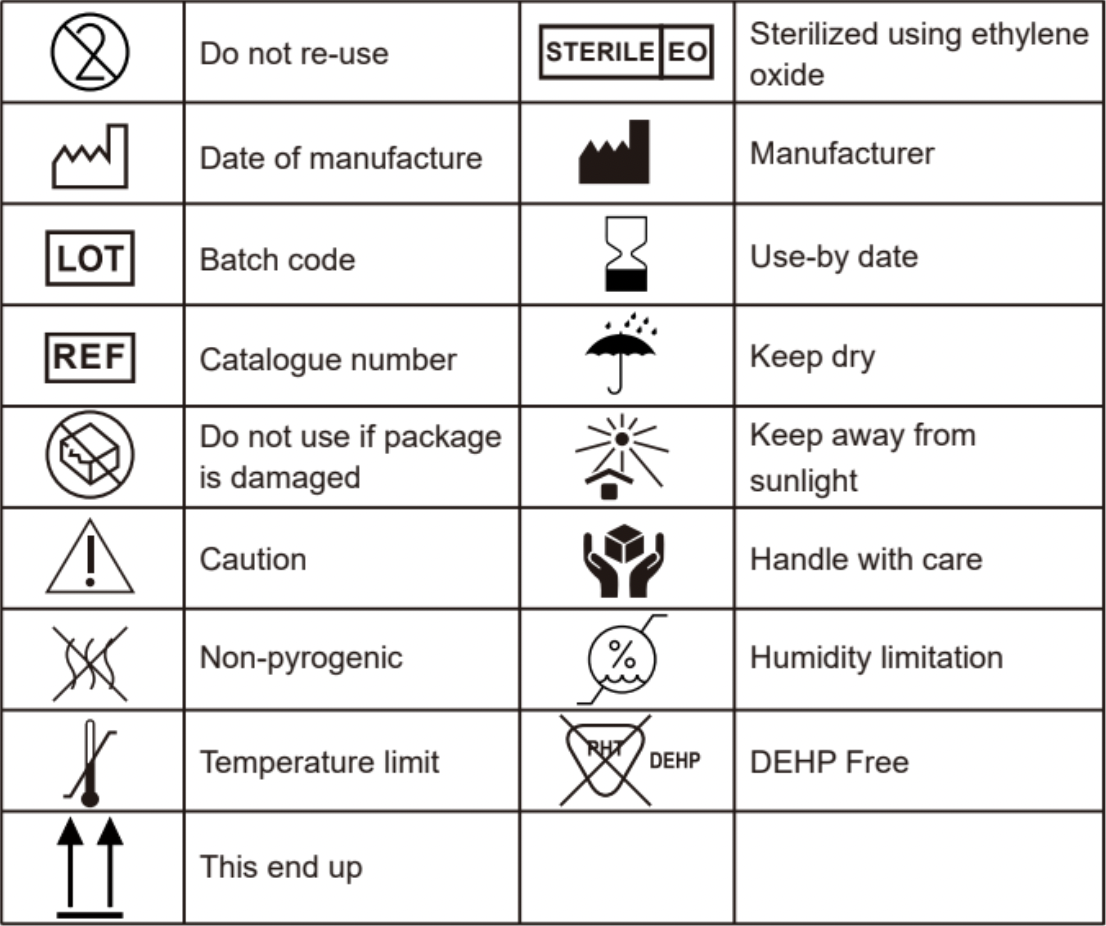
9. Symbol